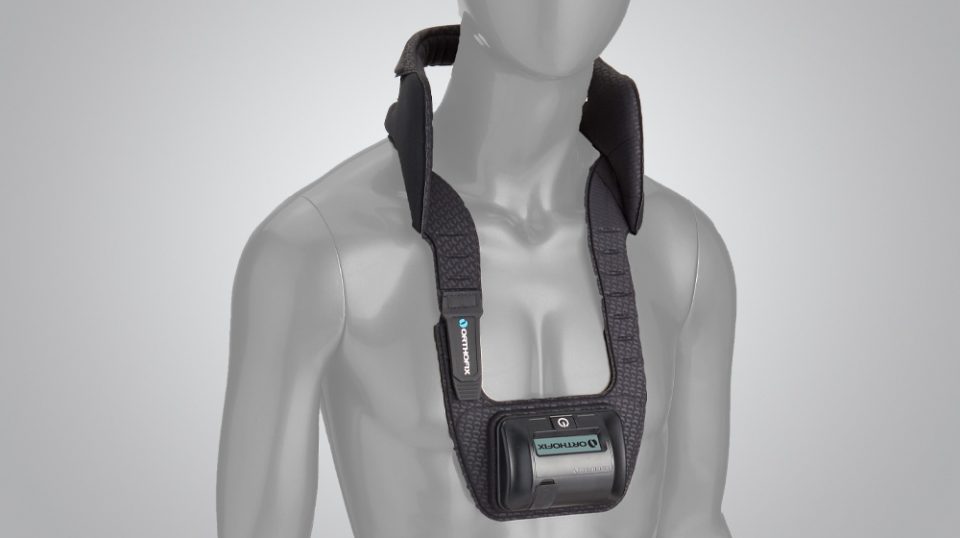
The CervicalStim device has been approved by the FDA to be worn after cervical spine fusion surgery in patients at risk for non-fusion.1,2 For complete prescribing information, please refer to the Instruction Manual.
It is a single-piece device that is lightweight, flexible and portable, allowing freedom of movement during treatment. An LCD and audible alarm provide important feedback during treatment such as the operational status, treatment time remaining, battery capacity, etc.
Designed for patient ease of use
- Works effectively when worn over clothing or bracing
- Single-piece, cordless design allows for ease of placement and patient mobility
- The STIM onTrack™mobile app is patient-friendly and provides patients with a treatment calendar, therapy reminders, and additional educational resources.*
This device utilizes Orthofix PEMF technology that provides 360 degrees of treatment coverage around the fusion site, covering up to five vertebral levels.3
Why do physicians prescribe a CervicalStim device?
- High clinical success rates1,2
- Statistically significant results for patients who smoke or have a multi-level fusion1,2
- PEMF signal covers 360 degrees around the fusion site3
- Coverage up to 5 vertebral levels3
- Supported by the North American Spine Society’s coverage recommendations4
- STIM on Track mobile app enables physicians to remotely view patient adherence to prescribed treatments
* STIM onTrack mobile app is available as an accessory for US model devices only